We have a new publication in Frontiers in Neuroscience, The AII Amacrine Cell Connectome: A Dense Network Hub. Authors are Robert E. Marc, James R. Anderson, myself, Crystal Sigulinsky and J. Scott Lauritzen.
Abstract: The mammalian AII retinal amacrine cell is a narrow-field, multistratified glycinergic neuron best known for its role in collecting scotopic signals from rod bipolar cells and distributing them to ON and OFF cone pathways in a crossover network via a combination of inhibitory synapses and heterocellular AII::ON cone bipolar cell gap junctions. Long considered a simple cell, a full connectomics analysis shows that AII cells possess the most complex interaction repertoire of any known vertebrate neuron, contacting at least 28 different cell classes, including every class of retinal bipolar cell. Beyond its basic role in distributing rod signals to cone pathways, the AII cell may also mediate narrow-field feedback and feedforward inhibition for the photopic OFF channel, photopic ON-OFF inhibitory crossover signaling, and serves as a nexus for a collection of inhibitory networks arising from cone pathways that likely negotiate fast switching between cone and rod vision. Further analysis of the complete synaptic counts for five AII cells shows that (1) synaptic sampling is normalized for anatomic target encounter rates; (2) qualitative targeting is specific and apparently errorless; and (3) that AII cells strongly differentiate partner cohorts by synaptic and/or coupling weights. The AII network is a dense hub connecting all primary retinal excitatory channels via precisely weighted drive and specific polarities. Homologs of AII amacrine cells have yet to be identified in non-mammalians, but we propose that such homologs should be narrow-field glycinergic amacrine cells driving photopic ON-OFF crossover via heterocellular coupling with ON cone bipolar cells and glycinergic synapses on OFF cone bipolar cells. The specific evolutionary event creating the mammalian AII scotopic-photopic hub would then simply be the emergence of large numbers of pure rod bipolar cells.
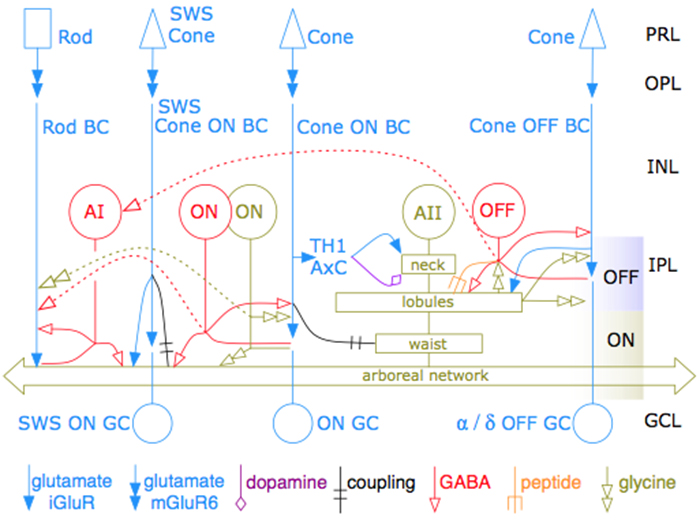
I know more about photography than retinal research but I am somewhat confused by the last sentence of the article. In order for evolution to produce the network hub described wouldn’t it also be necessary to develop the complex interconnecting network of communication that you have illustrated and described, or am I misunderstanding the article?
Not sure I understand your question. These networks have evolved over 300 million years, sometimes coming and going. The rod pathways in mammals are “piggybacked” onto the cone pathways through the AII amacrine cell.
Thanks for your reply Bryan. I apologize for any confusion my question may have caused. Please be patient with me since my knowledge of the subject is much more limited than yours. Let me start by saying that I understand the retina as being similar to an extremely complex interconnected electrical micro component system. I have seen the word circuitry used to describe the coordinated communication of the component cellular systems of the retina. When I think of circuitry I think of an electrical system.
As a result of seeing the retina as complex micro circuitry, I do not see the simple addition of new component parts to be the major challenge for the evolution of the retina. If a part is added to the system it must also be “re-wired” in an integrated and coordinated manner so the new part will function properly within the system and confer a new function without disrupting the existing “electrical circuitry”.
The question I was trying to ask was if the emergence of large numbers of pure rod bipolar cells alone would be adequate to explain the process you and the other authors described. In addition to the new cells wouldn’t a new “wiring schematic” also have to be developed? If so, I see that as a more difficult problem than simply adding the cells.
In regard to your answer, I wasn’t clear on what you meant by sometimes coming and going. Could you please explain in more detail what you meant by that.
No worries. I just did not understand what you were asking.
We are simply describing the circuitry and cell types that are present. The hypothesis would be that circuitry in the mammalian is similar across mammalian species. There may be some variation depending upon ecological niche, but many of the rules should be robust. Some more complex retinas like turtle for instance would have different circuitry reflecting their cell populations and ecological niche. A couple hundred million years of evolution are pretty powerful engines for circuit refinement.
As far as coming and going, I meant that vision has evolved several times through history. Some organisms lost their visual systems and eventually re-evolved them. It is thought that the rod system “piggybacked” onto an older cone-dominated system for instance.
Thanks again for your reply and your clarification. Please understand that I am not questioning your research in any way. I think the research you and the others are doing is very exciting and very well done. My only concern is about the qualification of a completely unguided process with no “wiring diagram or schematic” in hand to accomplish the development of what seems to be brilliantly executed retinal circuitry and function. When you say vision has evolved several times throughout history it seems even more incredulous to me. You and the other extremely intelligent researchers have spent many hours trying to understand one very small part of an extremely complex micro component system. I have a great deal of difficulty understanding how that system was produced without some type of plan or foresight.
The work of Mary Schweitzer and others makes me question the millions of years that have been proposed for evolutionary development. Her work with dinosaur soft tissues has been very enlightening. I have seen photomicrographs of what appear to be extremely well preserved nucleated red blood cells that were found within soft elastic structures that appear to be blood vessels. The first time I saw one particular photo I was actual startled by how similar it appeared to extant structures and cells of the same type. I know Dr Schweitzer’s group has proposed a possible solution to this dilemma but I have a great deal of difficulty with some of the assumptions her group has made in reaching the conclusions they have drawn about long term preservation of soft tissue. It would take an entirely new blog to cover that topic.
Even if we stay with the proposed millions of years for evolution I still have difficulty understanding the development of the retina through an unguided process. Natural selection cannot produce a construction plan of any sort. It can only eliminate what already exists. Evolution has to rely on mutation or mistakes in DNA editing to produce a new structure like the retina. I would be very interested in knowing if you or anyone in your group has an estimate for the number of mutations it would take to construct an advanced retina from scratch. Going through the process from no retina to the most complex retina we have today, how many beneficial mutations (since mutations are overwhelmingly neutral or slightly deleterious) would it take and how often could we expect to get one? If we had those numbers we could plug them in to a population genetics program and find the mathematical probability of developing the retina through an evolutionary process. When doing such calculations it would be necessary to also recognize that the mutations would have to be coordinated. By coordinated I mean confined to only one very tiny and specific area of the genome – the area that codes for the retina. My personal opinion is that we would find the mathematical odds against the evolution of the retina overwhelmingly stacked against it happening even once.
A 1985 article in Byte magazine compared the processing speed of the retina to a Cray supercomputer of that era. I know that computer technology has progressed considerably since that time, but so has our knowledge of the retina. If indeed the retina processes information like a computer then the retina would also require “reprogramming” many times during the evolutionary process. It would be a little like starting with a very early computer operating system and progressing to windows 8.1 without a computer engineer to program the changes. Again, my personal opinion is that the mathematical odds against producing an advanced retina in an undirected manner would make it astronomically improbable to happen even once.